 So-called "smokable hemp" is suddenly available in shops from coast to coast, legal under terms of last year's Farm Bill. But law enforcement is consternated, conservative politicians are apoplectic, and legislation is being prepared to ban the stuff at the state level. Is this permissible under federal law?
So-called "smokable hemp" is suddenly available in shops from coast to coast, legal under terms of last year's Farm Bill. But law enforcement is consternated, conservative politicians are apoplectic, and legislation is being prepared to ban the stuff at the state level. Is this permissible under federal law?

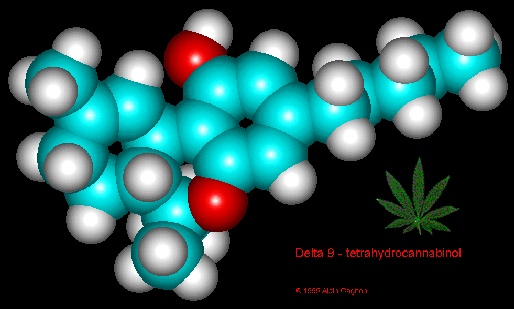 Researchers are exploring the connection between cannabinoids and dopamine in the human brain, and particularly how THC affects our "reward system"—key to such phenomena as motivation, addiction and euphoria. Investigations are revealing both potential for treatments of psychiatric disorders as well as possible risks from heavy use, especially among the young.
Researchers are exploring the connection between cannabinoids and dopamine in the human brain, and particularly how THC affects our "reward system"—key to such phenomena as motivation, addiction and euphoria. Investigations are revealing both potential for treatments of psychiatric disorders as well as possible risks from heavy use, especially among the young. With the fall of Italy’s far-right government, advocates are hoping the path may be cleared now for the country to become Europe's first to formally legalize cannabis. Meanwhile, thanks to a loophole in the law, low-THC varieties are sold openly in shops across the country. And Italy’s internal THC limit is actually slightly higher than the ultra-cautious European Union standard.
With the fall of Italy’s far-right government, advocates are hoping the path may be cleared now for the country to become Europe's first to formally legalize cannabis. Meanwhile, thanks to a loophole in the law, low-THC varieties are sold openly in shops across the country. And Italy’s internal THC limit is actually slightly higher than the ultra-cautious European Union standard.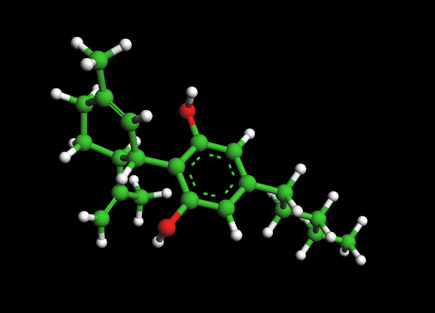 Among the multiple grim challenges facing humanity at this moment is the specter of "antibiotic apocalypse"—so-called "superbugs" developing resistance to common antibiotics, portending a plague of incurable infections. Research in Australia now reveals anti-bacterial properties in CBD, effective even against the growing ranks of resistant superbugs. Many in the stateside cannabis industry say the development is further evidence that legal barriers to research need to come down—and fast.
Among the multiple grim challenges facing humanity at this moment is the specter of "antibiotic apocalypse"—so-called "superbugs" developing resistance to common antibiotics, portending a plague of incurable infections. Research in Australia now reveals anti-bacterial properties in CBD, effective even against the growing ranks of resistant superbugs. Many in the stateside cannabis industry say the development is further evidence that legal barriers to research need to come down—and fast. The prosecutor for Miami-Dade County is the latest of several around the country to halt minor cannabis cases. The move was prompted by a dilemma vexing law enforcement nationwide: the inability to distinguish between THC and legal CBD in confiscated samples.
The prosecutor for Miami-Dade County is the latest of several around the country to halt minor cannabis cases. The move was prompted by a dilemma vexing law enforcement nationwide: the inability to distinguish between THC and legal CBD in confiscated samples. Hundreds of cannabis possession charges have been dismissed in Texas in recent weeks because police don't have labs that can differentiate between marijuana and newly legal hemp. The governor, attorney general and politicians are up in arms about it. Other states are updating their test kits to distinguish between CBD and THC in confiscated samples.
Hundreds of cannabis possession charges have been dismissed in Texas in recent weeks because police don't have labs that can differentiate between marijuana and newly legal hemp. The governor, attorney general and politicians are up in arms about it. Other states are updating their test kits to distinguish between CBD and THC in confiscated samples. Building on longstanding policy that bars federal cannabis enforcement in medical marijuana states, the House of Representatives passed a measure that would instate a similar hands-off approach to enforcement in states that have generally legalized. Other measures would slash funding for the DEA, and call upon the FDA to promulgate regs for CBD.
Building on longstanding policy that bars federal cannabis enforcement in medical marijuana states, the House of Representatives passed a measure that would instate a similar hands-off approach to enforcement in states that have generally legalized. Other measures would slash funding for the DEA, and call upon the FDA to promulgate regs for CBD.





Recent comments
3 weeks 2 days ago
4 weeks 3 days ago
4 weeks 3 days ago
5 weeks 5 days ago
11 weeks 2 hours ago
13 weeks 3 days ago
18 weeks 3 days ago
30 weeks 3 days ago
32 weeks 4 days ago
33 weeks 4 days ago